As I mentioned last week, I’ve been ill. For those who wanted the grisly details, well: here you go.
Warning: this blog post contains frank subjective descriptions of the symptoms of slightly-icky medical conditions including photographs. It’s probably safe for all audiences, but you might not want to be drinking anything while you read it.
The trouble began, I suppose, about a month and a half ago, when a contracted what seemed to be a minor urinary tract infection. If you’re a woman then, statistically-speaking, I probably don’t need to tell you what that is, but for the benefit of the men: it’s what happens when bacteria (or, sometimes, a fungus or virus) infects the renal system: the kidneys, bladder, ureters, and especially the urethra. It’s not pleasant: it gives you the feeling of needing to pee the whole time, makes it harder to pee, and – when you do – it feels a little bit like you’re piss is made of lava.
Despite it not being common for men (more on that later), I’ve had mild UTIs on a couple of occasions in my life, and I’d always found that ensuring that I got plenty of water and a full RDA of vitamin C was more than enough to make it clear up all by itself within a couple of days. So that’s what I started doing. But then things took a turn for the worse: I started getting a stabbing pain in my left kidney. Recognising this as being pyelonephritis, I went to the doctor who prescribed me a course of the antibiotic ciprofloxacin. Within a couple of days I was feeling right as rain (of course I continued to finish the course of drugs, although I was interested to see that that advice is starting to become controversial).

Naturally I was a little disappointed when, the week before last, I started getting UTI-like pain again, followed very swiftly this time by pain in my bladder that constantly felt a little like I was recovering from being punched. Back to the doctor I went, where (after the usual tests to work out what the most-likely best-antibiotic to use was) I was prescribed a course of nitrofurantoin. I’d never had this particular drug before, and it wasn’t initially clear which of the escalating ill-effects I was experiencing were symptoms of the infection and which were side-effects of the medication: it started with joint pain, then nausea, then diarrhoea, then a full-on fever. It was at the point that I was fully-clothed in bed, running a temperature and soaked in sweat but still feeling cold and shivering that Ruth called 111, who told her to take me to A&E.
(Which, like her care for me in general, she did fabulously well, except for a little bit where she sort-of ran me over in the car park of the hospital: thankfully some friendly paramedics were standing around and were able to drag me into the building. Anyway, I don’t have much memory of that bit and I certainly don’t have any amusing photos, so I’ll skip over it.)
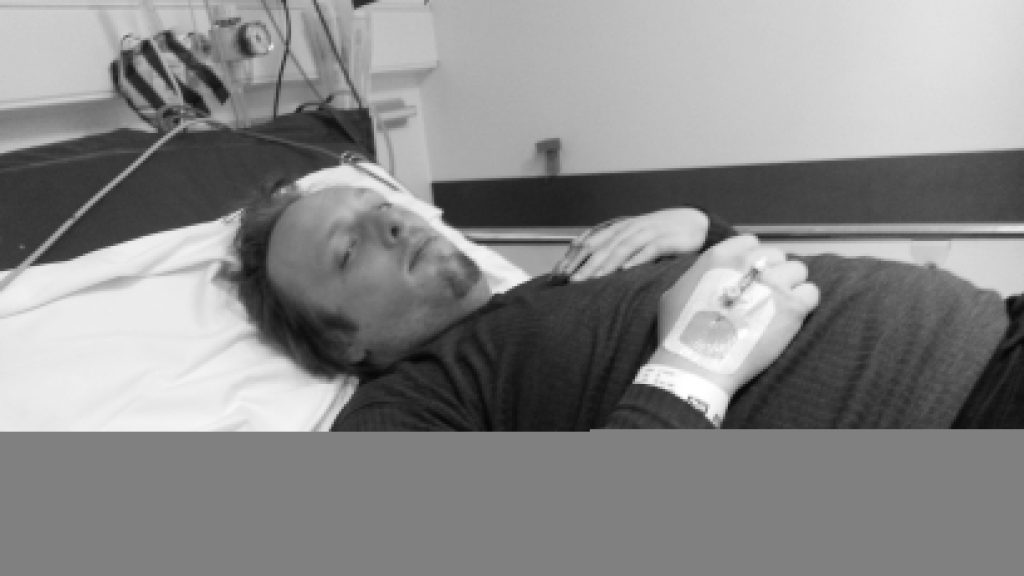
A few tests later, the medical staff seemed confident that what I was experiencing was not an allergic reaction to the antibiotic (however, I see that they still made a note of it as a risk on my notes!) but was a progression of the infection, which seemed to have crossed over from the tissues of my renal system and into my bloodstream and was now generally causing havoc by attacking the rest of my body. They hooked me up to a drip of an intravenous antibiotic and kept me stocked with painkillers, then sent me up to the urology ward and set me up with a “bed” (urology mostly deals with outpatients, and so my ‘bed’ was actually a trolley, but they wanted to keep me close to the urologists in case of any complications).

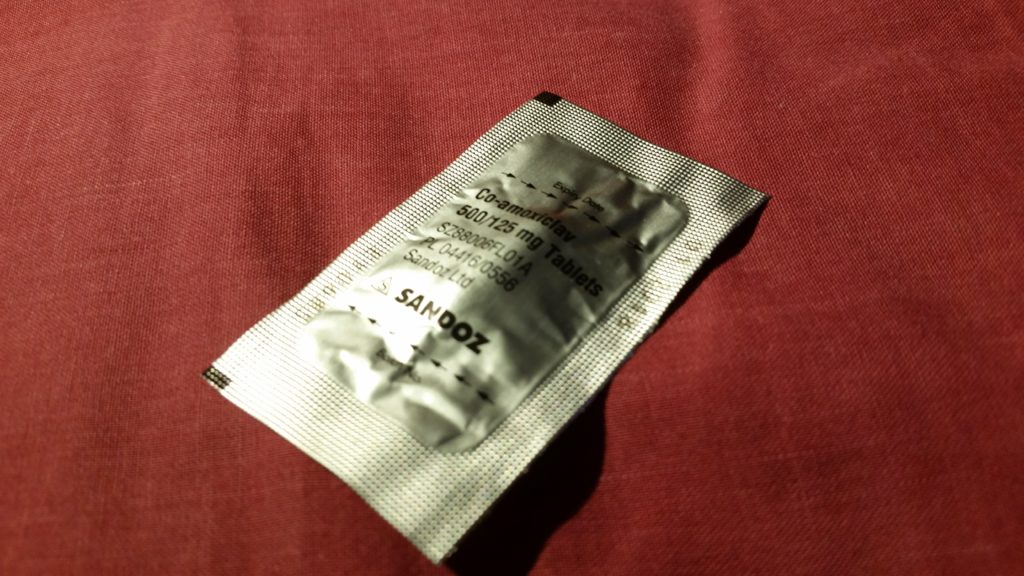
A consultant switched me to a week’s course of yet-another different antibiotic – co-amoxiclav – and recommended keeping me in for another night. Now, I think that co-amoxiclav is a really interesting drug, so I’m going to be a bit of a nerd and tell you about that for a bit (I promise we’ll get back to my health in a moment: if you don’t want the science bit, just scroll past the diagrams to the next photo).
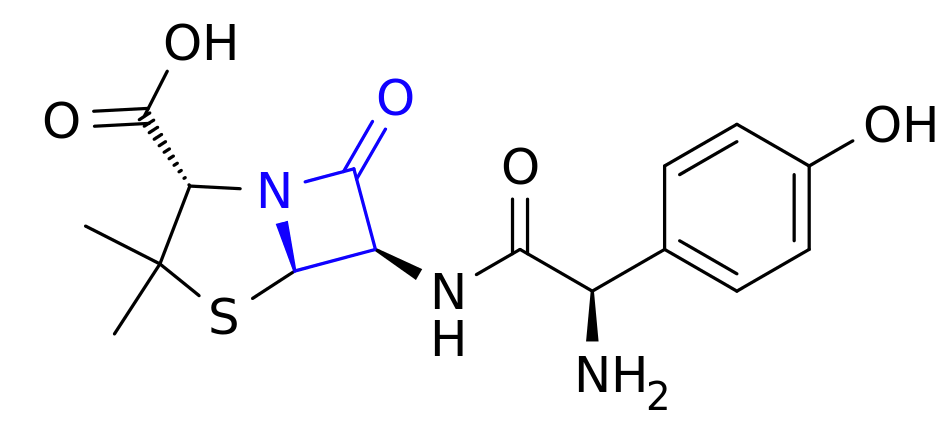
Co-amoxiclav is a mixture of two drugs. The first is the antibiotic amoxicillin. Amoxicillin belongs to a class of antibiotics (which includes penicillin) called β-lactams, which is the most-commonly used family of antibiotics. These antibiotics contain a four-point lactam ‘ring’ (highlighted in blue above), and the way that they work is that this part of the molecule bonds with a particular protein common to all gram-positive bacteria. Normally this protein is responsible for producing peptidoglycan, which is an essential ingredient in the cell walls of these kinds of bacteria, but when it gets locked to a β-lactam ring it stops working. As a result, when the bacterium reproduces the new child doesn’t have a proper cell wall, and can’t survive long in even the least-hostile environments.
Of course, we’re in a medical arms race right now because some of the bacteria which we’re targetting with antibiotics are becoming resistant. And here’s one what that they’re doing so: some of these bacteria have evolved to produce beta-lactamase, also bonds with beta-lactam rings, adding an OH to them and making them useless. Bummer, eh?
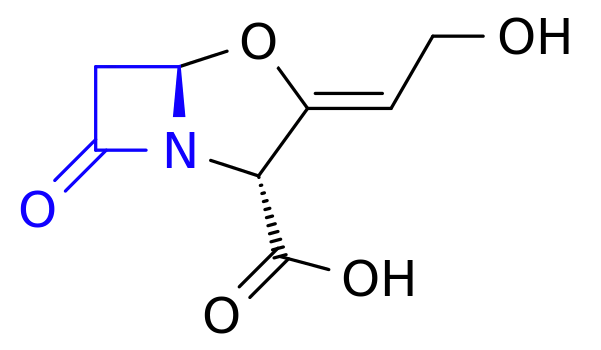
The second drug in co-amoxiclav, then, is clavulanic acid, which was discovered in the 1970s and started being added to drugs in the 1980s. Despite having a β-lactam ring (as you’ll see in blue above), clavulanic acid by itself it isn’t an effective antibiotic (for reasons I can’t quite get my head around – anyone want to help me?). But what it’s great at is bonding that lactam ring to beta-lactamase, thereby deactivating the bacterial counter-offensive and allowing the amoxicillin to carry on working, combating resistance.
So what you’ve got in co-amoxiclav is a an antibiotic and a chemical that counteracts the effects of a chemical that deactivates that antibiotic. Wow! It’s things like this that really make me wish I had a brain for biology!
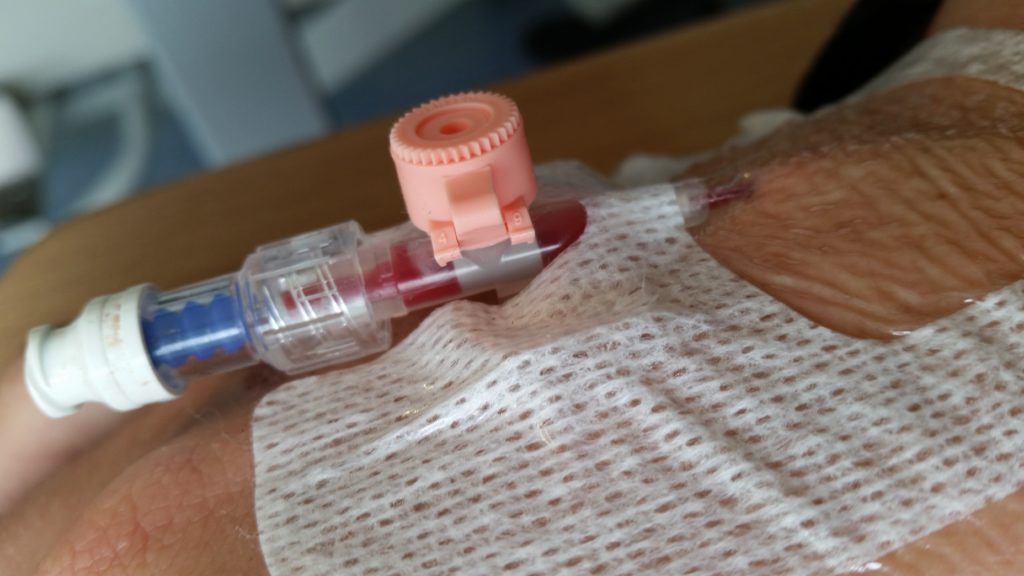
I was eventually discharged from hospital and released to go home for lots of bed rest and water, along with a further week’s course of co-amoxiclav. Unfortunately it turns out that I’m one of the unlucky folks for whom amoxicillin makes me dizzy, so I spent most of that week lying down in-between wobbly vertigo-filled trips to and from the bathroom. But it worked! Within a few days I was feeling much better and by the end of last week I was able to work from home (and actually feel like I was useful again!).
Free of symptoms and off the drugs, I returned to work properly on Monday morning and everything seemed fine. Until, late in the morning, I went to the bathroom and started pissing blood.

Now apparently blood in your urine, while horrifying when it happens to you unexpectedly, isn’t actually a sign of a medical emergency. I was starting to get bladder pain again, quite intensely, so I excused myself from work and called the urology ward, who decided that I wasn’t in bad enough a condition to go and see them but sent me straight to my GP, who gave me another fortnight’s worth of co-amoxiclav. They’re monitoring my progress with urine and blood samples and if by Friday it’s not having an impact, they’re going to want to send me back to hospital (hopefully only as an outpatient) and pump me full of the intravenous stuff again. So… fingers crossed for a good result out of these drugs.
I was hoping that by this point I’d be writing this blog post and telling you all about how I’d fought the bladder monster and won. But it looks like I won’t be able to claim that victory for another week or two, yet. All I know is that I searched for “bladder monster” and found this. Yeah: that feels about right.
Well that’s a nice blog there… Lemme just bookmark that one.
Sorry to hear you are not well just yet but you are the right tract at least!
If you think that you can get away with making puns like that then urine for a surprise.
@scatmandan @AlecofWharf my brain autocompleted with “and the bladder monster won!” D:
Oh man, I hope that they determine it is nothing and you continue on your recovery!
That really does not sound like fun. (Also the car part)
We sure have missed you these past few days.
UTIs are no joking matter. They can kill you pretty quick. I get them a lot too, and the doctors give me a week’s worth of Bactrim that knocks it out in a day or two.
The cold chills while sweating profusely fucking sucks. Sorry for the bad language, but that warrants the emphatic interjection.
I’m guessing the pills are in a packet under Nitrogen because some portion of the molecule or binding agent will oxidize.
I’ve not had a UTI, and you make me hope I never do!
While they’re a lot more-common in women (statistically-speaking, most women will suffer from one at least once during their life – traditionally, often “honeymoon cystitis” shortly after they first become sexually-active with a male partner), they’re apparently a lot more-likely to result in complications when they’re contracted by men.
Ah. I wonder if that’s because men tend to “tough it out” and women go to the doctor at the first sign of pain?
I think it’s mostly to do with anatomical differences. Womens’ shorter urethra and the closer proximity of the urethral opening to the anus makes it more-likely that bacteria will enter the urethra and reach the bladder. Plus there’s the factor of the second-biggest risk group: recently menopausal women, whose hormone changes can result in variation in vulval flora, which can create an environment in which various yeasts and bacteria can thrive. (Aren’t you glad you asked!)
Meanwhile in men the longer urethra results in more exposed area for infection… and if the prostate becomes inflamed then this can put further pressure on the bladder and make it harder still to empty completely, which makes it harder for the body to flush out the invaders.
That’s my understanding of it, anyway. Here’s more, from the NHS.
Your anatomical explanation makes sense…but I still prefer my answer!
I didn’t run you over! You started falling out of the car, and then I unfortunately stalled it while jumping out to come and help. The door hit you. A little bit.
Sounds awful, get well soon!
@scatmandan poor bugger. But, welcome to the injured junk club Geniteer!
Three weeks ago was (give or take a few weeks because we’ve never bothered with accuracy) the end of Ruth and I’s 8th year together, and we marked the occasion with a mini-break away for a few nights. We spent the first two nights in a ‘showman’-style gypsy caravan in Herefordshire, and it was amazing enough that I wanted to share it with you:
It wasn’t quite dusk yet, but we couldn’t resist the urge to light the fire (and the dozens of tiny lanterns).The place we went was Wriggles Brook, a ‘glamping’-style site in the shadow of the Forest of Dean. In a long field that twists its way alongside a babbling brook, the owners have set up a trio of traditional horse-drawn caravans, each in a wooded clearing that isolates it from the others. Two of the caravans are smaller, designed just for couples (who are clearly the target market for this romantic getaway spot), but we took the third, larger, (centenarian!) one, which sported a separate living room and bedroom.
Between our caravan and the others the owners grew a varied orchard, which Annabel found particularly interesting. By which I mean delicious.The bedroom was set up so that children could be accommodated in a bunk under the adults (with their own string of fairy lights and teeny-tiny windows, but after she bumped her head on the underside of the beams Annabel decided that she didn’t want to sleep there, so we set up her travel cot in the living room.
Annabel and I swinging on the hammock near theSo yeah: a beautiful setting, imaginative and ecologically-friendly accommodation, and about a billion activities on your doorstep. Even the almost-complete lack of phone signal into the valley was pretty delightful, although it did make consulting Google Maps difficult when we got lost about 20 minutes out from the place! But if there’s one thing that really does deserve extra-special mention, it’s the food!
Nearby activities include steam trains. That’s all I needed to hear, really.Our hosts were able to put on a spectacular breakfast and evening meal for us each night, including a variety of freshly-grown produce from their own land. We generally ate in their mini dining room – itself a greenhouse for their grapevines – but it was equally-nice to have pancakes delivered to the picnic table right outside our caravan. And speaking as somebody who’s had their fair share of second-rate veggie breakfasts over the last… what, four and a half years?… it was a great relief to enjoy a quite-brilliant variety of vegetarian cuisine from a clearly-talented chef.
I’m not sure why the Wriggles Brook site has ocassional signs like this sticking out of the undergrowth, but they sort-of fit the eccentricity of the place.So yeah – five stars for Wriggles Brook in Herefordshire if you’re looking for an awesome romantic getaway, with or without an accompanying toddler. Ruth and I later palmed the little one off on JTA so that we could have a night away without her, too, which – while fun (even if we didn’t get to try all 280+ gins at the restaurant we ate at) – wasn’t quite so worthy of mention as the unusual gypsy-caravan-escape that had preceded it. I’m hoping that we’ll get out to Wriggles Brook again.
Read more →
Warning: this blog post contains pictures of urine, invasive equipment, and the inside of a bladder. It’s probably safe for all audiences, but you might like to put your glass of apple juice down for a minute or two. The short of it all is that I’m probably healthy.
Since my hospitalisation the other month with a renal system infection, I’ve undergone a series of investigations to try to determine if there’s an underlying reason that I fell ill. As my doctor explained to me, it’s quite possible that what I’d experienced was a random opportunistic infection (perhaps aided by a course of unrelated antibiotics I’d been on earlier this year or by certain lifestyle habits), but if that wasn’t the case – if there were some deeper explanation for my health problems – it was important to find out sooner, rather than later.
I’ve peed in so many little pots! If you laid them end-to-end across your kitchen counter, people would think that you were some kind of pervert.Early on I had several ultrasound scans of my bladder (at a number of different times and at a variety of levels of fullness) and one of my kidneys, the latter of which revealed some “minor scarring” of one of them which apparently isn’t something I should be worried about… although I wish they’d started the two-page letter I got with that rather than opening with, effectively, “Contrary to what we told you at the hospital, we did later see something wrong with you…” But still, good to be reassured that this is probably not an issue.
An ultrasound scan of one of my kidneys. Can you tell the sex yet?More recently, I went to the hospital to have a “flow rate test” and a cystoscopy. The flow rate test involved the most-ghetto looking piece of NHS equipment I’ve ever seen: functionally, it seemed to be little more than a funnel on top of a large measuring beaker, in turn on top of a pressure-sensitive digital scale. The scale was connected up to the only fancy-looking bit of equipment in the room, a graphing printer that output the calculated volume (based on their weight) of the same and, more-importantly, the rate of change: the “flow rate” of the stream of urine.
I’m right, aren’t I? That’s basically a kitchen funnel, isn’t it?I suppose one advantage of using equipment like this is that it basically operates itself. Which meant that the nurse was able to give me five seconds worth of instruction and then leave the room, which saved us from our own Britishness forcing us to make small-talk while I urinated in front of her or something. Ultimately, I turned out to be within the range of normalcy here, too, although I was a little disappointed to find that the ward didn’t maintain a daily “score board” of flow rates, as sort-of a science-backed literal pissing contest.
Apparently not all men experience that ‘spurt-and-then-full-pressure’ thing you’ll see on the graph on the right, when they start to pee, but some of us do, and it’s perfectly normal. I’m learning so much!Finally came the cystoscopy, and this was the bit that I’d been most-nervous about. This procedure involves the insertion of a long flexible tube into the urethra at the tip of the penis, under local anasthetic, and pushing it all the way down, through the sphincter, down through the prostate and then back up into the bladder. It’s then used as a channel to pump water into the bladder, filling it to capacity and stretching out the sides, after which the fibreoptic cord (and light) that runs along its length is used to look around inside the bladder to inspect for any of a plethora of different problems.
You’re going to put that WHERE?The doctor invited me to watch with him on the monitor, which I initially assumed was because I was clearly interested in everything and kept asking questions, but in hindsight I wonder if it’s just that he – quite rightly – assumed that I might have panicked if I’d have been looking in the direction of the piece of equipment he brought in and jabbed at my penis with. I only looked at it while it was on its way out, and my god its a scary-looking thing: sort of like a cross between a tyre pressure gauge and a blowtorch. The first few inches were painless – the local anasthetic had made me completely numb right up to and including the external sphincter, which is at the base of the penis. However, what I can only assume was the second sphincter complained of the discomfort, and it stung pretty sharply any time the doctor would twist the cystoscope to change the angle of the picture.
The view as you ‘travel’ up the urethra looks pretty much like I expected. With a motion simulator, it would make a pretty cool ride!Seeing the inside of your own body is an amazing experience. I mean: it’s not amazing enough to even be worth the experience of a cystoscopy, never mind the illness that in my case preceeded it… but it’s still pretty cool. The ultrasounds were interesting, but there’s nothing quite so immersive as seeing a picture of the inside of your own bladder, gritting your teeth while the doctor points to an indentation and explains that it’s the opening to the ureter that connects to your own left kidney!
Unfortunately I neglected to take my phone into the operating room, having put it into a locker when I changed into a gown, and so I wasn’t able to (as I’d hoped) take photos of the inside of my own bladder. So you’ll have to make do with this video I found, which approximates the experience pretty well. The good news is that there’s probably nothing wrong with me, now that the infection from earlier this year has passed: nothing to suggest that there’s any deeper underlying issue that caused me to get sick, anyway!
The bad news is that while the procedure itself was shorter and more-bearable than I’d expected, the recovery’s been a real drag. A week later, it still hurts a lot to urinate (although I’ve stopped yelping out loud when I do so) and my crotch is still too sore for me to be able to cycle. I’ve also discovered that an errection can be painful enough to wake me up, which is definitely not the most-pleasant way I’ve been roused by a penis. But it’s getting better, day by day, and at least I know for sure that I’m more-or-less “right” in the renal system, now.
Read more →