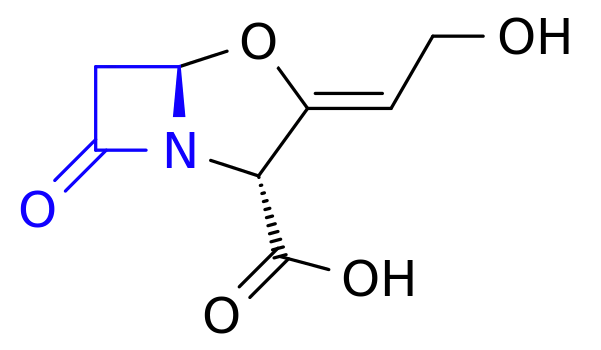
Warning: this blog post contains pictures of urine, invasive equipment, and the inside of a bladder. It’s probably safe for all audiences, but you might like to put your glass of apple juice down for a minute or two. The short of it all is that I’m probably healthy.
Since my hospitalisation the other month with a renal system infection, I’ve undergone a series of investigations to try to determine if there’s an underlying reason that I fell ill. As my doctor explained to me, it’s quite possible that what I’d experienced was a random opportunistic infection (perhaps aided by a course of unrelated antibiotics I’d been on earlier this year or by certain lifestyle habits), but if that wasn’t the case – if there were some deeper explanation for my health problems – it was important to find out sooner, rather than later.

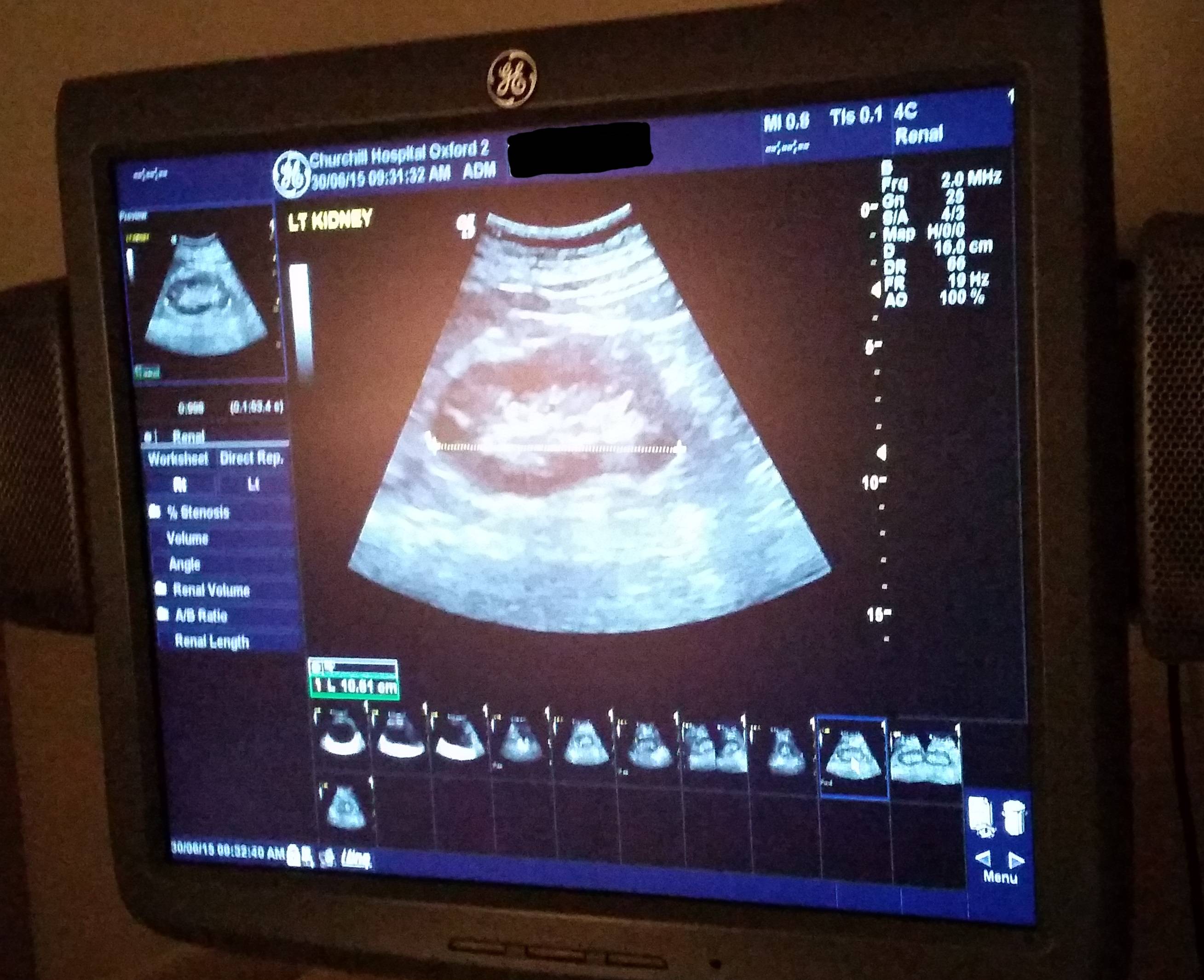
Early on I had several ultrasound scans of my bladder (at a number of different times and at a variety of levels of fullness) and one of my kidneys, the latter of which revealed some “minor scarring” of one of them which apparently isn’t something I should be worried about… although I wish they’d started the two-page letter I got with that rather than opening with, effectively, “Contrary to what we told you at the hospital, we did later see something wrong with you…” But still, good to be reassured that this is probably not an issue.

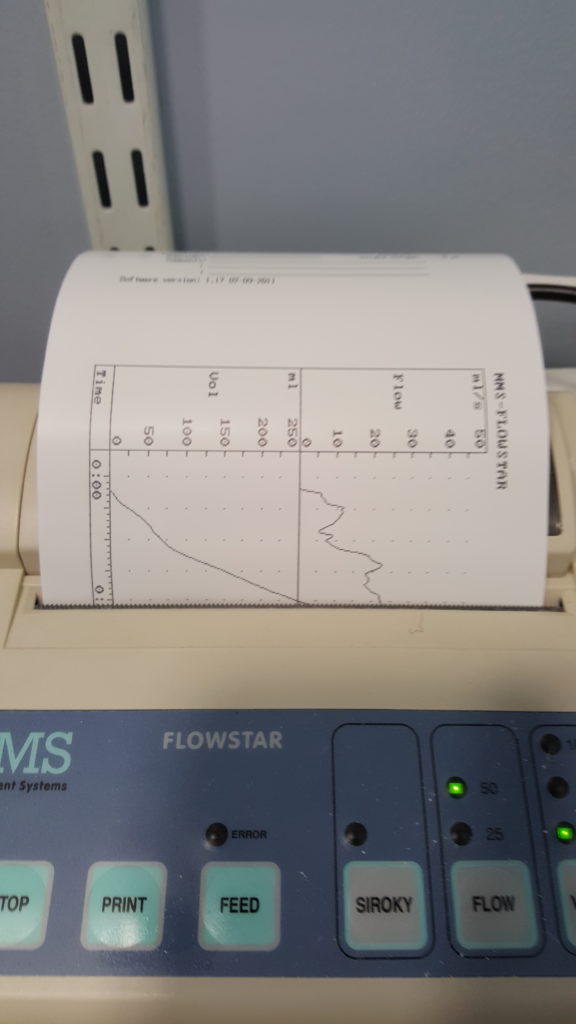
More recently, I went to the hospital to have a “flow rate test” and a cystoscopy. The flow rate test involved the most-ghetto looking piece of NHS equipment I’ve ever seen: functionally, it seemed to be little more than a funnel on top of a large measuring beaker, in turn on top of a pressure-sensitive digital scale. The scale was connected up to the only fancy-looking bit of equipment in the room, a graphing printer that output the calculated volume (based on their weight) of the same and, more-importantly, the rate of change: the “flow rate” of the stream of urine.

I suppose one advantage of using equipment like this is that it basically operates itself. Which meant that the nurse was able to give me five seconds worth of instruction and then leave the room, which saved us from our own Britishness forcing us to make small-talk while I urinated in front of her or something. Ultimately, I turned out to be within the range of normalcy here, too, although I was a little disappointed to find that the ward didn’t maintain a daily “score board” of flow rates, as sort-of a science-backed literal pissing contest.
Finally came the cystoscopy, and this was the bit that I’d been most-nervous about. This procedure involves the insertion of a long flexible tube into the urethra at the tip of the penis, under local anasthetic, and pushing it all the way down, through the sphincter, down through the prostate and then back up into the bladder. It’s then used as a channel to pump water into the bladder, filling it to capacity and stretching out the sides, after which the fibreoptic cord (and light) that runs along its length is used to look around inside the bladder to inspect for any of a plethora of different problems.
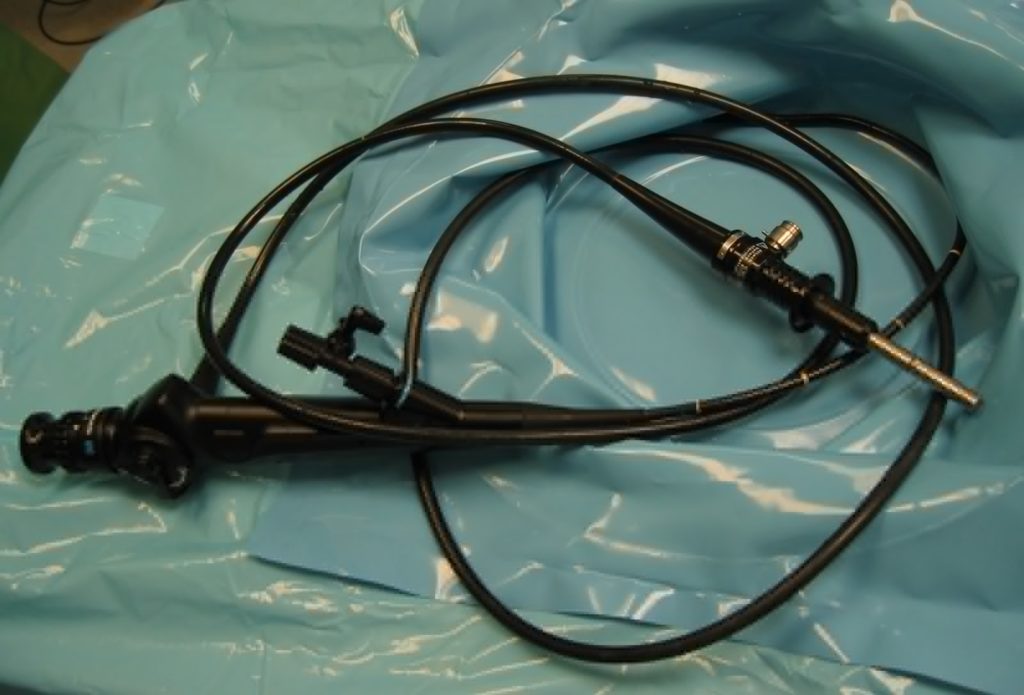
The doctor invited me to watch with him on the monitor, which I initially assumed was because I was clearly interested in everything and kept asking questions, but in hindsight I wonder if it’s just that he – quite rightly – assumed that I might have panicked if I’d have been looking in the direction of the piece of equipment he brought in and jabbed at my penis with. I only looked at it while it was on its way out, and my god its a scary-looking thing: sort of like a cross between a tyre pressure gauge and a blowtorch. The first few inches were painless – the local anasthetic had made me completely numb right up to and including the external sphincter, which is at the base of the penis. However, what I can only assume was the second sphincter complained of the discomfort, and it stung pretty sharply any time the doctor would twist the cystoscope to change the angle of the picture.
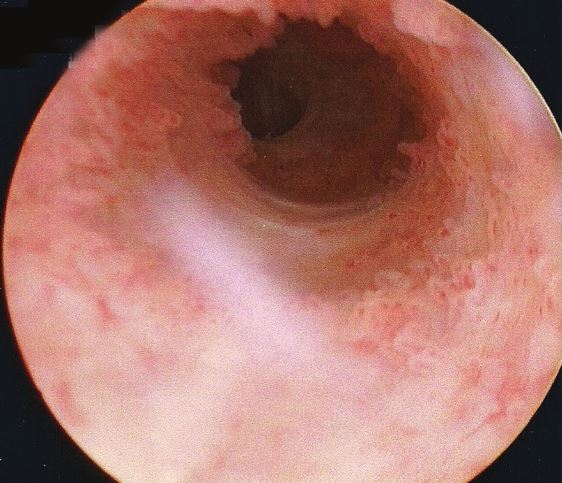
Seeing the inside of your own body is an amazing experience. I mean: it’s not amazing enough to even be worth the experience of a cystoscopy, never mind the illness that in my case preceeded it… but it’s still pretty cool. The ultrasounds were interesting, but there’s nothing quite so immersive as seeing a picture of the inside of your own bladder, gritting your teeth while the doctor points to an indentation and explains that it’s the opening to the ureter that connects to your own left kidney!
Unfortunately I neglected to take my phone into the operating room, having put it into a locker when I changed into a gown, and so I wasn’t able to (as I’d hoped) take photos of the inside of my own bladder. So you’ll have to make do with this video I found, which approximates the experience pretty well. The good news is that there’s probably nothing wrong with me, now that the infection from earlier this year has passed: nothing to suggest that there’s any deeper underlying issue that caused me to get sick, anyway!
The bad news is that while the procedure itself was shorter and more-bearable than I’d expected, the recovery’s been a real drag. A week later, it still hurts a lot to urinate (although I’ve stopped yelping out loud when I do so) and my crotch is still too sore for me to be able to cycle. I’ve also discovered that an errection can be painful enough to wake me up, which is definitely not the most-pleasant way I’ve been roused by a penis. But it’s getting better, day by day, and at least I know for sure that I’m more-or-less “right” in the renal system, now.